Good Manufacturing Practice (or GMP) comprises guidelines on quality assurance that govern the manufacturing processes and environments for a vast range of goods including medicinal products and medical devices. The goal is a high-quality production process all the way from material procurement through to warehouse logistics: the key to high product quality. But above and beyond this, quality management compliant with GMP and ISO 13485 should ensure that the regulatory requirements for marketing the products are met. In other words: validated high product quality is actually an essential condition in the medtech industry, rather than mainly a competitive advantage or differentiating feature as is the case in other industries.
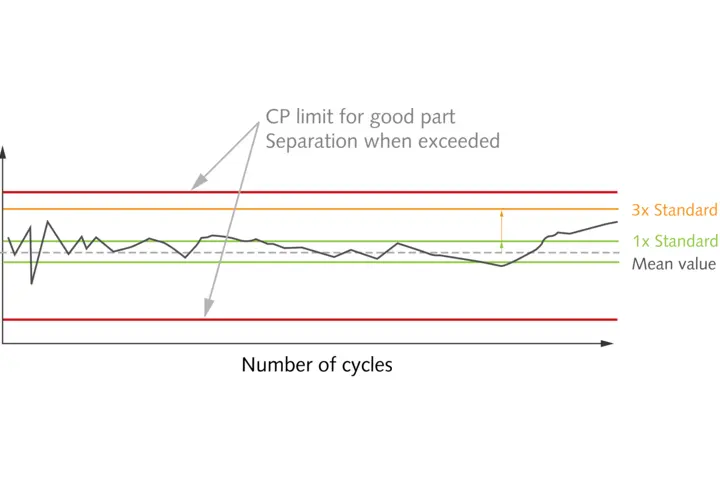
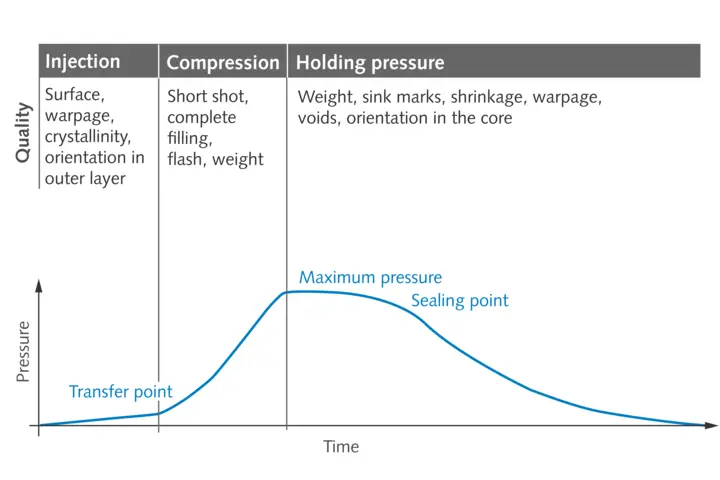
The two core elements of GMP are qualification of machinery and plant, and validation of processes and methods. The first element requires a planned and documented multi-stage process to show that plant and equipment are basically suitable for the purpose, and that they do actually function reliably under the conditions prevailing on site. Process and method validation also calls for documentation and proof that the processes and methods used will generate reliable and reproducible results, and that the manufactured product will conform to the requirements. To achieve these high standards – especially as regards documentation – process monitoring based on cavity pressure has repeatedly proven its effectiveness in injection molding practice. This parameter is the most informative process value in injection molding, because it gives users a completely transparent view of the process to create a molded part. It goes without saying that this helps users to meet their documentation obligations. And as added benefits, cavity pressure monitoring simplifies process validation during machine setup and makes it easier to optimize production processes – with zero-defect production based on quality prediction models as the ultimate goal.